Prescription drug makers are cutting back on marketing for opioid-related payouts to doctors, according to an analysis by ProPublica.
Big Pharma payed more than $15 million to doctors for opioid marketing, which was 33 percent less than the year before.
These figures come as the latest update to the nonprofit’s Dollars for Docs online tool. It tracks the payments to doctors from drug and medical companies.
The latest numbers show that the $15 million for doctors in exchange for opioid-related speaking and consulting. It is 21 percent less than the 2014 figure. It has steadily dropped in 2015 and 2016.
Research has drawn a link between marketing and prescribing opioids to patients.
Assistant Professor of Health Policy and Management at Harvard told ProPublica that if this the manufacturers are being aware of their role in the opioid epidemic, it’s a move in the right direction.
A growing number of lawsuits against drug companies, who are accused of downplaying the risks of prescription opioid painkillers in their marketing campaigns and contributed to the opioid crisis.
Although the marketing contributes to the problem, experts say it’s not all that is driving the increased numbers of opioid use.
However, when the marketing dollars decreased, the number of opioid prescriptions started decreasing as well. The dip in marketing funds has outpaced the reduction in prescriptions.
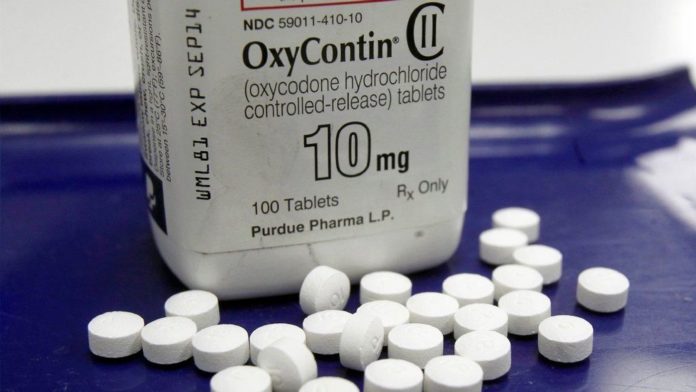
Purdue Pharma, the producer of OxyContin, cut off its speaker program for the opioid in the year 2016. This year, the company stopped all of their physician-target promotional efforts of its addictive prescription painkillers and laid off some of their sales representatives.
The company said last month that “While the development of new medicines will be the company’s priority going forward,” they would continue to support the opioid analgesic product portfolio while continuing their commitment to take meaningful steps necessary to reduce opioid abuse and dependence.
OxyContin was passed by the FDA in 1995 and since then it has been Purdue Pharma’s biggest success, even amid the rise of generic alternatives and growing popularity of other opioids.